|
This follows the reactivity trend for group 7 Halogens any halogen will displace halide ions from aqueous solution of any members of the group below it in the Periodic Table. Alli, MD on NovemWhats a Vaccine Which Ones Does My Child Need 7 Through 18 Years Old Why So Many Shots at Once Why Is My Child Getting The Same. Step 4 was repeated with bromine water and chlorine water as shown in the movie clip.Ĭolour changes due to displacement reactions were observed (are clearly visible in the movie clip).Ĭhlorine displaces both bromide and iodide ions from solution.īromine displaces iodide ions from solution. A few millilitres of aqueous iodine was added to one flask of potassium chloride, one flask of potassium bromide and one flask of potassium iodide and swirled.ĥ. 20ml potassium iodide was put into three 100ml conical flasks.Ĥ. 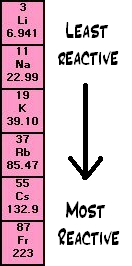
20ml potassium bromide was put into three 100ml conical flasks.ģ. Approximately 20ml potassium chloride was put into three 100ml conical flasks.Ģ. Explaining the Reactivity Trend of the Group 7 Halogen. describe and explain displacement reactions between group 7 elements. explain the trend in reactivity in group 7. predict the properties of other group 7 elements. describe trends in physical properties of these elements. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
